 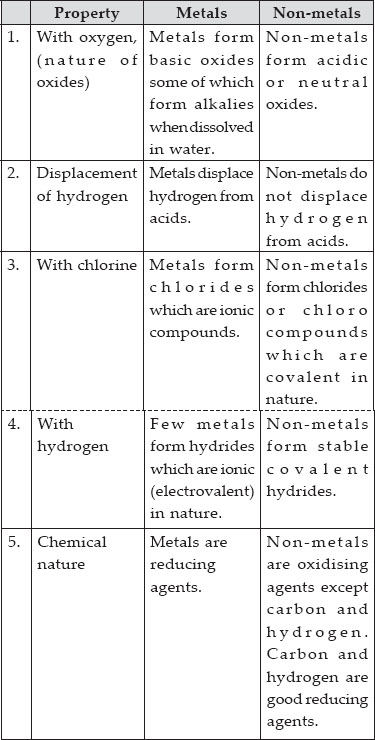
The second equation shows us that tin is more active then copper. Does the following reaction occur Mg (s) CuCl2(aq) MgCl2(aq) Cu (s) Answer. A metal listed above another metal will replace that metal in a single replacement (single displacement) reaction. In the first reaction we determined that zinc is more active than copper and the third reaction shows us that zinc is also more active than tin. Answer: The metal reactivity series lists metals in order of their reactivity, from highest to lowest from top to bottom. Using all three of the reactions above, we can make an activity series for Cu, Zn, and Tin. If a piece of zinc is placed in the SnCl 2 a reaction occurs. 
In this case the tin is more active than the copper, as the copper is not able to force its electrons onto the tin. Suppose a piece of copper was placed in a solution of SnCl 2, and nothing happens. When an ion keeps the same charge in a reaction and is just there to balance out the opposite charges, we call it a Spectator Ion. 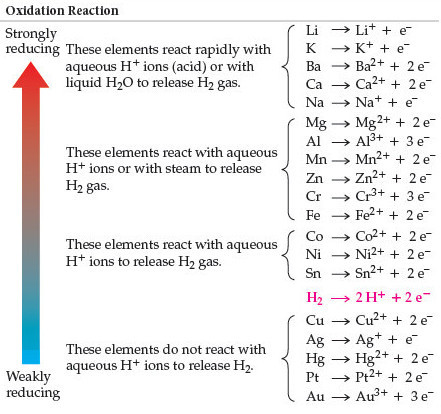
Nitrate ion in both the reactants and the products has a -1 charge. The reactivity of a metal is related to its tendency (how likely it is) to form. In the equation below, nitrate is there to balance the charges. When metals react with other substances the metal atoms form positive ions. Positive ions (cations) can't just be floating around by themselves in solution, they have to have a negative ion (anion) to balance them. We say that Zn is more active than the copper. From properties to structures, everything you need to help your 1416 students grasp metals and alloys. If Al is above Co in the activity series of metals, which of the following will occur if Al metal is put into a solution of cobalt nitrate answer choices. It does not store any personal data.\): An activity series for common metals.Įxample: Zn and Cu both tend to lose 2 electrons, but the zinc wants to lose them more, so much so that it can force the two electrons on Cu 2 and make it become Cu again. bubbles of oxygen gas will form on iron nail. The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. Activity Series of Metals: The electrochemical, electromotive, or activity series of the elements is formed when the electrodes (metals and nonmetals) in contact with their ions are ordered on the basis of the values of their standard reduction potentials or standard oxidation potentials. The cookie is used to store the user consent for the cookies in the category "Performance". Activity Series: An easy way to use the table is to note that a metal can reduce any ion of a metal below it in the table. This cookie is set by GDPR Cookie Consent plugin. The ranking of metals (and hydrogen) based on their activity is called the activity series. The reactivity series of metals is a list of metals arranged in their order of reactivity from highest to lowest. be higher on the activity seriesthan the metal cation in solution. The cookie is used to store the user consent for the cookies in the category "Other. The activity series is shown below: Li > K > Ba > Sr > Ca > Na > Mg > Al > Mn > Zn > Fe > Cd > Co > Ni > Sn > Pb > (H) > Cu > Ag > Hg > Au For a single-replacement reaction to occur, the free metal must be more activei.e. This cookie is set by GDPR Cookie Consent plugin. Reactivity Series of Metals - Reactivity series, extracting metals - Reactivity of Metals - Reaction of metals with water and acid - reactivity series. The cookies is used to store the user consent for the cookies in the category "Necessary". 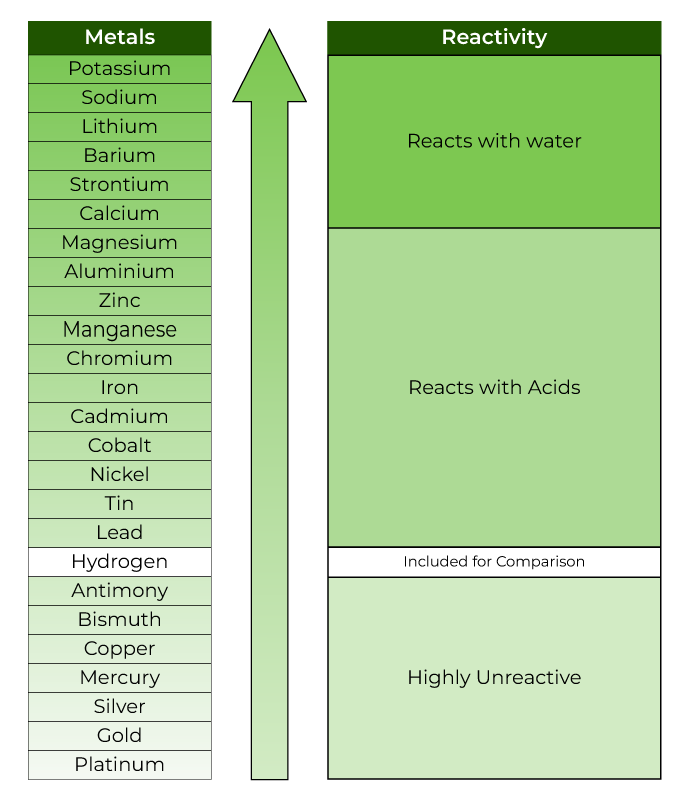
This cookie is set by GDPR Cookie Consent plugin. The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". If it loses electrons really easily, it is really reactive, and so goes at the. These cookies ensure basic functionalities and security features of the website, anonymously. For metals, the reactivity of a metal depends on how easily it loses electrons. Necessary cookies are absolutely essential for the website to function properly. Typically, in order to determine the reactivity series, the metals would be measured for their reactivity against a reactant like water, acid, or another. The metals placed above hydrogen in the series can displace it from acids such as HCl and H 2SO 4 (since they are more reactive). Persulfate-based advanced oxidation processes have been widely praised in the treatment of organic contaminants, but the intrinsic factors of peroxymonosulfate (PMS) activation have not been identified. H (Non-Metal, Reference for Comparison)ĭespite being a non-metal, hydrogen is often included in the reactivity series since it helps compare the reactivities of the metals. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
